Carbon nanotube
In 1991 Iijima Sumio of NEC Corporation’s Fundamental Research Laboratory, Tsukuba Science City, Japan, investigated material extracted from solids that grew on the tips of carbon electrodes after being discharged under C60 formation conditions. Iijima found that the solids consisted of tiny tubes made up of numerous concentric “graphene” cylinders, each cylinder wall consisting of a sheet of carbon atoms arranged in hexagonal rings.

The cylinders usually had closed-off ends and ranged from 2 to 10 micrometres (millionths of a metre) in length and 5 to 40 nanometres (billionths of a metre) in diameter. High-resolution transmission electron microscopy later revealed that these multiwalled carbon nanotubes (MWNTs) are seamless and that the spacings between adjacent layers is about 0.34 nanometre, close to the spacing observed between sheets of graphite. The number of concentric cylinders in a given tube ranged from 3 to 50, and the ends were generally capped by fullerene domes that included pentagonal rings (necessary for closure of the tubes). It was soon shown that single-walled nanotubes (SWNTs) could be produced by this method if a cobalt-nickel catalyst was used. In 1996 a group led by Smalley produced SWNTs in high purity by laser vaporization of carbon impregnated with cobalt and nickel. These nanotubes are essentially elongated fullerenes.

Individual carbon nanotubes may be metallic or semiconducting, depending on the helical orientation of the rows of hexagonal rings in the walls of the tubes. Rather than conducting electricity via electron transport, a diffusive process that results in electron scattering and conductive heating, SWNTs exhibit ballistic transport, a highly efficient and fast conduction process in which electrons, prevented from diffusing through the wall of the tube or around its circumference by the regular hexagonal array of carbon atoms, propagate rapidly along the axis of the tube. Open-ended SWNTs emit electrons at currents that attain approximately 100 nanoamperes (billionths of an ampere). Owing to such remarkable properties, electrical conductors made of bundles of nanotubes should exhibit zero energy loss. Aligned MWNTs show promise as field-emission devices with potential applications in electronic flat-panel displays. Nanotubes may also be used as highly resilient probe tips for scanning tunneling microscopes and atomic force microscopes.
Carbon nanotubes exhibit faster phonon transport than diamond, which was previously recognized as the best thermal conductor, and the electric current-carrying capacity of nanotubes is approximately four orders of magnitude higher than that of copper. The Young’s modulus of MWNTs (a measure of their elasticity, or ability to recover from stretching or compression) is estimated by researchers to be greater than that of carbon fibres by a factor of 5 to 10. MWNTs are capable of readily absorbing loads via a sequence of reversible elastic deformations, such as buckling or kinking, in which the bonds between carbon atoms remain intact.

Nanotubes can be “decapped” by oxidation and the resulting opened tubes filled with metals, such as lead, or even with buckyballs. Boron and nitrogen atoms may be incorporated into carbon nanotube walls. Microscopic metal particles that would otherwise be rapidly oxidized may be stabilized in air by encapsulation in nanotube skins.
Since then, carbon nanotubes with various structures have been discovered. According to the number of graphic shells, they are mainly categorized as single-walled (SWNTs) and multi-walled carbon nanotubes (MWNTs). The carbon nanotubes reported by Iijima were MWNTs synthesized by arc discharge methods. Two years later, two sets of researchers working independently—Iijima and Toshinari Ichihashi, along with Donald S. Bethune and his colleagues at IBM—synthesized SWNTs, using transition-metal catalyzed arc discharge.

A SWNT can be described as a long tube formed by wrapping a single graphene sheet into a cylinder with diameter of about 1 nanometer, the ends of which are capped by fullerene cages. The fullerene structures, with alternating structures of five hexagons adjacent to one pentagon, form the surface with desired curvature to enclose the volume. The sidewalls of carbon nanotubes are made of graphene sheets consisting of neighboring hexagonal cells. Other polygon structures, such as pentagons and heptagons, constitute defects of sidewalls. The cylindrical sidewalls can be produced from different rolling directions to make SWNTs with distinct structures and properties. Due to cylindrical symmetry, there are only a handful of methods that are effective in making seamless cylinders, and they are characterized by the chiral vectors with integer indices (n, m).
To establish the chiral vector, two atoms in the graphene sheet are selected, with one serving as the origin of the vector pointing toward the other atom. The graphene sheet is then rolled in a way that allows the two atoms to coincide. Under these circumstances, the chiral vectors form a plane perpendicular to the longitude direction of nanotubes and the lengths of the chiral vectors are equal to the circumference. Three different types of SWNTs are distinctly characterized, named “zigzag” (m = 0), “armchair” (n = m), and “chiral.” These structural variations result in differences in electrical conductivity and mechanical strength.
MWNTs are concentrically aligned SWNT assemblies with different diameters. The distance between adjacent shells is about 0.34 nanometer. MWNTs differ from SWNTs not only in their dimensions, but also in their corresponding properties. Various techniques have been developed to produce carbon nanotubes in sizable quantity, high yield, and purity, while maintaining a reasonable cost. Well-developed techniques include arc discharge, laser ablation, and chemical vapor deposition (CVD), and most processes involve costly vacuum conditions.
Arc discharge was initially used for the synthesis of fullerenes. In a typical experimental setup, a chamber filled with low-pressure (50 to 700 mbar) inert gas (helium, argon) is where the reaction takes place. Two carbon rods are placed end-to-end as the electrodes, separated by a few millimeters, and a direct current of 50 to 100 A (driven by a potential difference of 20 V) generates a high discharge temperature to sublime the negative electrode, leaving soot where carbon nanotubes are found. This method is the most common way to synthesize carbon nanotubes and perhaps the easiest way. The quality of carbon nanotubes depends on the uniformity of plasma arc, catalysts, and the selection of filling gases. A mixture of carbon nanotubes is usually produced; thus, purification processes are needed to remove the fullerenes, amorphous carbon, and catalysts.

Laser ablation was first employed to produce carbon nanotubes in 1995. A pulsed or continuous laser is used to vaporize a graphite (or graphite metal mixture) target in a 1,200 °C (2,200 °F) oven filled with inert gas at a pressure of 500 torr. Carbon vapors cool down rapidly during expansion, and carbon atoms quickly condense to form tubular structures with the help of catalyst particles. MWNTs can be synthesized when pure graphite is vaporized, and SWNTs are grown from graphite-transition metal (cobalt, nickel, etc.) mixtures. The method is primarily used to synthesize SWNTs with high selectivity and in a diameter-controllable way by tailoring reaction temperatures. The resulting products are usually in the form of bundles. Laser ablation is the most costly technique due to the involvement of expensive lasers and high power input.
Chemical vapor deposition (CVD) is the most promising way to produce carbon nanotubes on an industrial scale. This process utilizes high energy (600–900 °C [1,100–1,650 °F]) to atomize gaseous carbon sources, such as methane, carbon monoxide, and acetylene. The resulting reactive carbon atoms diffuse toward a catalyst-coated substrate and condense to form carbon nanotubes. Well-aligned carbon nanotubes can be synthesized with precisely controlled morphology, provided that proper reaction conditions are maintained, including preparation of substrates, selection of catalysts, etc.
Novel chemical, electrical, and mechanical properties absent in other materials have been discovered in carbon nanotubes. Pristine carbon nanotubes are inert to most chemicals and need to be grafted with surface functional groups to increase their chemical reactivity and add new properties. For SWNTs, electrical conductivity is dependent on the chiral vector and independent of the length as determined by quantum mechanics. Considering a chiral vector with indices (n, m), carbon nanotubes are metallic when n = m or (n - m) = 3i (i is an integer) and semiconducting in other cases. Along the longitude directions, carbon nanotubes show superior mechanical strength, with the highest known tensile strength and elastic modulus among known materials.
As for thermal properties, carbon nanotubes outperform diamond as the best thermal conductor. Applications of carbon nanotubes are aimed to make use of their unique properties to solve problems at the nanoscale. Their high surface area, together with the unique ability to carry any chemical compounds after surface modification, offers carbon nanotubes the potential to be used as nanoscale catalyst supports with high catalytic reactivity and chemical sensors. They are known to be the best field emitters due to their sharp tips, which can concentrate electric field easily, enabling them to emit electrons at low voltages.
This property has special applications in field emission flat-panel displays and cold-cathode electron guns used in microscopes. In nanoelectronics, SWNTs have been used to fabricate transistors that can function at room temperature and are potential candidates for devices operating at tetrahertz (THZ) frequencies. Engineering materials using carbon nanotubes as additives have exhibited capability to make plastic composites with enhanced electrical conductivity and mechanical strength. For biomedical applications, carbon nanotubes show promise as vehicles for targeted drug-delivery and nerve cell regeneration. However, their future success in bio-related applications is highly subject to the toxicity study, which is still in an early stage.
Some researchers have become concerned about the health risks involving carbon nanotubes, which according to lab research seem to pose a danger to human health that is similar to asbestos. In particular, exposure to carbon nanotubes has been associated with mesothelioma, a cancer of the lung lining. If inhaled, it is believed that nanotubes can scar lung tissues in a manner similar to asbestos fibers, a cause for concern because nanotubes are already used in many common products, such as bicycle frames, automobile bodies, and tennis rackets. Potential health risks are relevant not only to those involved in manufacturing but also to the general public, and little research has been conducted to determine if risks to human health are created when products containing nanotubes are crushed or incinerated in a waste dump.
Properties
Mechanical
Carbon nanotubes are the strongest and stiffest materials yet discovered in terms of tensile strength and elastic modulus respectively. This strength results from the covalent sp2 bonds formed between the individual carbon atoms. In 2000, a multi-walled carbon nanotube was tested to have a tensile strength of 63 gigapascals (9,100,000 psi). (For illustration, this translates into the ability to endure tension of a weight equivalent to 6,422 kilograms-force (62,980 N; 14,160 lbf) on a cable with cross-section of 1 square millimetre (0.0016 sq in).) Further studies, such as one conducted in 2008, revealed that individual CNT shells have strengths of up to ≈100 gigapascals (15,000,000 psi), which is in agreement with quantum/atomistic models. Since carbon nanotubes have a low density for a solid of 1.3 to 1.4 g/cm3, its specific strength of up to 48,000 kN·m·kg−1 is the best of known materials, compared to high-carbon steel's 154 kN·m·kg−1.
Although the strength of individual CNT shells is extremely high, weak shear interactions between adjacent shells and tubes lead to significant reduction in the effective strength of multi-walled carbon nanotubes and carbon nanotube bundles down to only a few GPa. This limitation has been recently addressed by applying high-energy electron irradiation, which crosslinks inner shells and tubes, and effectively increases the strength of these materials to ≈60 GPa for multi-walled carbon nanotubes and ≈17 GPa for double-walled carbon nanotube bundles. CNTs are not nearly as strong under compression. Because of their hollow structure and high aspect ratio, they tend to undergo buckling when placed under compressive, torsional, or bending stress.
On the other hand, there was evidence that in the radial direction they are rather soft. The first transmission electron microscope observation of radial elasticity suggested that even the van der Waals forces can deform two adjacent nanotubes. Later, nanoindentations with atomic force microscope were performed by several groups to quantitatively measure radial elasticity of multiwalled carbon nanotubes and tapping/contact mode atomic force microscopy was also performed on single-walled carbon nanotubes. Young's modulus of on the order of several GPa showed that CNTs are in fact very soft in the radial direction.
Electrical
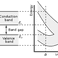
Unlike graphene, which is a two-dimensional semimetal, carbon nanotubes are either metallic or semiconducting along the tubular axis. For a given (n,m) nanotube, if n = m, the nanotube is metallic; if n − m is a multiple of 3 and n ≠ m and nm ≠ 0, then the nanotube is quasi-metallic with a very small band gap, otherwise the nanotube is a moderate semiconductor. Thus all armchair (n = m) nanotubes are metallic, and nanotubes (6,4), (9,1), etc. are semiconducting. Carbon nanotubes are not semimetallic because the degenerate point (that point where the π [bonding] band meets the π* [anti-bonding] band, at which the energy goes to zero) is slightly shifted away from the K point in the Brillouin zone due to the curvature of the tube surface, causing hybridization between the σ* and π* anti-bonding bands, modifying the band dispersion.
The rule regarding metallic versus semiconductor behavior has exceptions, because curvature effects in small diameter tubes can strongly influence electrical properties. Thus, a (5,0) SWCNT that should be semiconducting in fact is metallic according to the calculations. Likewise, zigzag and chiral SWCNTs with small diameters that should be metallic have a finite gap (armchair nanotubes remain metallic). In theory, metallic nanotubes can carry an electric current density of 4 × 109 A/cm2, which is more than 1,000 times greater than those of metals such as copper, where for copper interconnects current densities are limited by electromigration. Carbon nanotubes are thus being explored as interconnects, conductivity enhancing components in composite materials and many groups are attempting to commercialize highly conducting electrical wire assembled from individual carbon nanotubes. There are significant challenges to be overcome, however, such as undesired current saturation under voltage, the much more resistive nanotube-to-nanotube junctions and impurities, all of which lower the electrical conductivity of the macroscopic nanotube wires by orders of magnitude, as compared to the conductivity of the individual nanotubes.
Because of its nanoscale cross-section, electrons propagate only along the tube's axis. As a result, carbon nanotubes are frequently referred to as one-dimensional conductors. The maximum electrical conductance of a single-walled carbon nanotube is 2G0, where G0 = 2e2/h is the conductance of a single ballistic quantum channel.
Due to the role of the π-electron system in determining the electronic properties of graphene, doping in carbon nanotubes differs from that of bulk crystalline semiconductors from the same group of the periodic table (e.g. silicon). Graphitic substitution of carbon atoms in the nanotube wall by boron or nitrogen dopants leads to p-type and n-type behavior, respectively, as would be expected in silicon. However, some non-substitutional (intercalated or adsorbed) dopants introduced into a carbon nanotube, such as alkali metals as well as electron-rich metallocenes, result in n-type conduction because they donate electrons to the π-electron system of the nanotube. By contrast, π-electron acceptors such as FeCl3 or electron-deficient metallocenes function as p-type dopants since they draw π-electrons away from the top of the valence band.
Intrinsic superconductivity has been reported, although other experiments found no evidence of this, leaving the claim a subject of debate.
Optical
Carbon nanotubes have useful absorption, photoluminescence (fluorescence), and Raman spectroscopy properties. Spectroscopic methods offer the possibility of quick and non-destructive characterization of relatively large amounts of carbon nanotubes. There is a strong demand for such characterization from the industrial point of view: numerous parameters of the nanotube synthesis can be changed, intentionally or unintentionally, to alter the nanotube quality. As shown below, optical absorption, photoluminescence and Raman spectroscopies allow quick and reliable characterization of this "nanotube quality" in terms of non-tubular carbon content, structure (chirality) of the produced nanotubes, and structural defects. Those features determine nearly any other properties such as optical, mechanical, and electrical properties.
Carbon nanotubes are unique "one-dimensional systems" which can be envisioned as rolled single sheets of graphite (or more precisely graphene). This rolling can be done at different angles and curvatures resulting in different nanotube properties. The diameter typically varies in the range 0.4–40 nm (i.e. "only" ~100 times), but the length can vary ~100,000,000,000 times, from 0.14 nm to 55.5 cm. The nanotube aspect ratio, or the length-to-diameter ratio, can be as high as 132,000,000:1, which is unequalled by any other material. Consequently, all the properties of the carbon nanotubes relative to those of typical semiconductors are extremely anisotropic (directionally dependent) and tunable.
Whereas mechanical, electrical and electrochemical (supercapacitor) properties of the carbon nanotubes are well established and have immediate applications, the practical use of optical properties is yet unclear. The aforementioned tunability of properties is potentially useful in optics and photonics. In particular, light-emitting diodes (LEDs) and photo-detectors based on a single nanotube have been produced in the lab. Their unique feature is not the efficiency, which is yet relatively low, but the narrow selectivity in the wavelength of emission and detection of light and the possibility of its fine tuning through the nanotube structure. In addition, bolometer and optoelectronic memory devices have been realised on ensembles of single-walled carbon nanotubes.
Crystallographic defects also affect the tube's electrical properties. A common result is lowered conductivity through the defective region of the tube. A defect in armchair-type tubes (which can conduct electricity) can cause the surrounding region to become semiconducting, and single monatomic vacancies induce magnetic properties.
Thermal
All nanotubes are expected to be very good thermal conductors along the tube, exhibiting a property known as "ballistic conduction", but good insulators lateral to the tube axis. Measurements show that an individual SWNT has a room-temperature thermal conductivity along its axis of about 3500 W·m−1·K−1; compare this to copper, a metal well known for its good thermal conductivity, which transmits 385 W·m−1·K−1. An individual SWNT has a room-temperature thermal conductivity across its axis (in the radial direction) of about 1.52 W·m−1·K−1, which is about as thermally conductive as soil. Macroscopic assemblies of nanotubes such as films or fibres have reached up to 1500 W·m−1·K−1 so far. The temperature stability of carbon nanotubes is estimated to be up to 2800 °C in vacuum and about 750 °C in air.
Crystallographic defects strongly affect the tube's thermal properties. Such defects lead to phonon scattering, which in turn increases the relaxation rate of the phonons. This reduces the mean free path and reduces the thermal conductivity of nanotube structures. Phonon transport simulations indicate that substitutional defects such as nitrogen or boron will primarily lead to scattering of high-frequency optical phonons. However, larger-scale defects such as Stone Wales defects cause phonon scattering over a wide range of frequencies, leading to a greater reduction in thermal conductivity.
Single-shell carbon nanotubes of 1-nm diameterSimulation: Adhesion of Carbon Nanotubes




REFERENCES
Massachusetts Institute of Technology. Available in: http://web.mit.edu/mbuehler/www/SIMS/Adhesion%20of%20Carbon%20Nanotube.html. Access in: 28/10/2018.
Encyclopædia Britannica. Available in: https://www.britannica.com/science/carbon-nanotube. Access in: 28/10/2018.
Wikipedia. Available in: https://en.wikipedia.org/wiki/Carbon_nanotube#Properties. Access in: 28/10/2018.




























0 comments
Sign in or create a free account