Wave–particle duality
The de Broglie hypothesis, formulated in 1924, predicts that particles should also behave as waves. De Broglie's formula was confirmed three years later for electrons (which have a rest-mass) with the observation of electron diffraction in two independent experiments. George Paget Thomson, son of J. J. Thomson, passed a beam of electrons through a thin metal film and observed the predicted interference patterns. Around the same time at Bell Labs, Clinton Joseph Davisson and Lester Halbert Germer guided their beam through a crystalline grid.
The Electron diffraction refers to the wave nature of electrons. However, from a technical or practical point of view, it may be regarded as a technique used to study matter by firing electrons at a sample and observing the resulting interference pattern. This phenomenon is commonly known as wave–particle duality, which states that a particle of matter (in this case the incident electron) can be described as a wave. For this reason, an electron can be regarded as a wave much like sound or water waves.

Now that Schrödinger has described the matter wave, one of the phenomena we can study to understand how to interpret the wave function is diffraction. We know that a wave is subject to interference phenomena, whether constructive or destructive.
In relation to interference, in the case of classical wave mechanics, we can see from Young's experiment of double-slit diffraction that there are regions of constructive interference and total destructive interference,
If it were made with classical particles, the effect would not be seen, because the particle could only pass through 1 of the slits, unlike the wave that the wave front passes through the two slits at the same time. For the electron, we have to have a slit smaller than the size of the electron, which is complicated.
For a wave emitter and a particle emitter at a velocity as small as possible so that one particle reaches the slots at a time, and for a detector that picks up the intensity at which the wave or particle undergoes diffraction as it passes through the slit and stores the information of the accumulated intensities of the two slits as the following illustration.
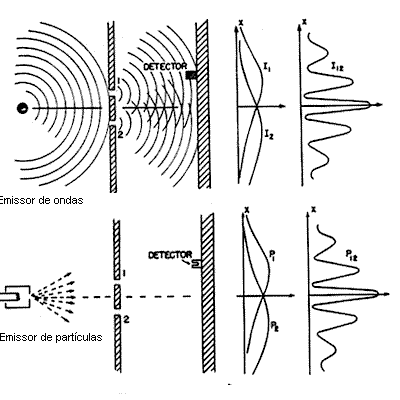
The following characteristics are observed:
- Particle Characteristics: There is always an integer number of electrons and an electron is emitted each time it reaches a point of the image at random;
- Wave characteristic: the accumulation of the effect gets fringes of interference; closing one of the apertures and closing one of the slits the detection may either decrease or increase depending on the emission position.
The electrons and other atomic and sub atomic particles are neither particles nor waves from the classical understanding, they present behaviors of each one of the classifications, but they are of nonclassic nature.



REFERENCES
NUSSENZVEIG, H. Moysés. Curso de Física Básica: Ótica, Relatividade, Física Quântica. vol. 4, 1ª ed. Editora Bluncher, São Paulo, 1998. p. 283-287.
Encyclopædia Britannica. Available in: https://www.britannica.com/science/wave-particle-duality. Access in: 07/09/2018.
Wikipedia. Available in: https://en.wikipedia.org/wiki/Electron_diffraction. Access in: 07/09/2018.




















0 comments
Sign in or create a free account