Alpha decay theory
Alpha particles were first described in the investigations of radioactivity by Ernest Rutherford in 1899, and by 1907 they were identified as He2+ ions.
By 1928, George Gamow had solved the theory of alpha decay via tunneling. The alpha particle is trapped in a potential well by the nucleus. Classically, it is forbidden to escape, but according to the (then) newly discovered principles of quantum mechanics, it has a tiny (but non-zero) probability of "tunneling" through the barrier and appearing on the other side to escape the nucleus. Gamow solved a model potential for the nucleus and derived, from first principles, a relationship between the half-life of the decay, and the energy of the emission, which had been previously discovered empirically, and was known as the Geiger–Nuttall law.

Alpha decay, type of radioactive disintegration in which some unstable atomic nuclei dissipate excess energy by spontaneously ejecting an alpha particle. Because alpha particles have two positive charges and a mass of four units, their emission from nuclei produces daughter nuclei having a positive nuclear charge or atomic number two units less than their parents and a mass of four units less.
The speed and hence the energy of an alpha particle ejected from a given nucleus is a specific property of the parent nucleus and determines the characteristic range or distance the alpha particle travels. Though ejected at speeds of about one-tenth that of light, alpha particles are not very penetrating.
For example, uranium-238 decays to form thorium-234. Alpha particles have a charge +2 e, but as a nuclear equation describes a nuclear reaction without considering the electrons – a convention that does not imply that the nuclei necessarily occur in neutral atoms – the charge is not usually shown.
Alpha decay typically occurs in the heaviest nuclides. Theoretically, it can occur only in nuclei somewhat heavier than nickel (element 28), where the overall binding energy per nucleon is no longer a minimum and the nuclides are therefore unstable toward spontaneous fission-type processes. In practice, this mode of decay has only been observed in nuclides considerably heavier than nickel, with the lightest known alpha emitters being the lightest isotopes (mass numbers 106–110) of tellurium (element 52). Exceptionally, however, beryllium-8 decays to two alpha particles.
Alpha decay is by far the most common form of cluster decay, where the parent atom ejects a defined daughter collection of nucleons, leaving another defined product behind. It is the most common form because of the combined extremely high binding energy and relatively small mass of the alpha particle. Like other cluster decays, alpha decay is fundamentally a quantum tunneling process. Unlike beta decay, it is governed by the interplay between both the nuclear force and the electromagnetic force.
The nuclear force holding an atomic nucleus together is very strong, in general much stronger than the repulsive electromagnetic forces between the protons. However, the nuclear force is also short range, dropping quickly in strength beyond about 1 femtometre, while the electromagnetic force has unlimited range. The strength of the attractive nuclear force keeping a nucleus together is thus proportional to the number of nucleons, but the total disruptive electromagnetic force trying to break the nucleus apart is roughly proportional to the square of its atomic number. A nucleus with 210 or more nucleons is so large that the strong nuclear force holding it together can just barely counterbalance the electromagnetic repulsion between the protons it contains. Alpha decay occurs in such nuclei as a means of increasing stability by reducing size.
One curiosity is why alpha particles, helium nuclei, should be preferentially emitted as opposed to other particles like a single proton or neutron or other atomic nuclei. Part of the answer comes from conservation of wave function symmetry, which prevents a particle from spontaneously changing from exhibiting Bose–Einstein statistics (if it had an even number of nucleons) to Fermi–Dirac statistics (if it had an odd number of nucleons) or vice versa. Single proton emission, or the emission of any particle with an odd number of nucleons would violate this conservation law. The rest of the answer comes from the very high binding energy of the alpha particle. Computing the total disintegration energy given by the equation:
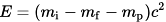
Where mi is the initial mass of the nucleus, mf is the mass of the nucleus after particle emission, and mp is the mass of the emitted particle, shows that alpha particle emission will usually be possible just with energy from the nucleus itself, while other decay modes will require additional energy. Most of this disintegration energy becomes the kinetic energy of the alpha particle itself, although to preserve conservation of momentum part of this energy becomes the recoil of the nucleus itself. However, since the mass numbers of most alpha emitting radioisotopes exceed 210, far greater than the mass number of the alpha particle (4) the part of the energy going to the recoil of the nucleus is generally quite small.
These disintegration energies however are substantially smaller than the potential barrier provided by the nuclear force, which prevents the alpha particle from escaping. The energy needed is generally in the range of about 25 MeV, the amount of work that must be done against electromagnetic repulsion to bring an alpha particle from infinity to a point near the nucleus just outside the range of the nuclear force's influence. An alpha particle can be thought of as being inside a potential barrier whose walls are 25 MeV. However, decay alpha particles only have kinetic energies of 4 MeV to about 9 MeV, far less than the energy needed to escape.
Quantum mechanics, however, provides a ready explanation, via the mechanism of quantum tunnelling. The quantum tunnelling theory of alpha decay, independently developed by George Gamow and Ronald Wilfred Gurney and Edward Condon in 1928, was hailed as a very striking confirmation of quantum theory. Essentially, the alpha particle escapes from the nucleus by quantum tunnelling its way out. Gurney and Condon made the following observation in their paper on it:
It has hitherto been necessary to postulate some special arbitrary ‘instability’ of the nucleus; but in the following note it is pointed out that disintegration is a natural consequence of the laws of quantum mechanics without any special hypothesis... Much has been written of the explosive violence with which the α-particle is hurled from its place in the nucleus. But from the process pictured above, one would rather say that the α-particle almost slips away unnoticed.
The theory supposes that the alpha particle can be considered an independent particle within a nucleus that is in constant motion, but held within the nucleus by nuclear forces. At each collision with the potential barrier of the nuclear force, there is a small non-zero probability that it will tunnel its way out. An alpha particle with a speed of 1.5×107 m/s within a nuclear diameter of approximately 10−14 m will collide with the barrier more than 1021 times per second. However if the probability of escape at each collision is very small, the half-life of the radioisotope will be very long, since it is the time required for the total probability of escape to reach 50%.
Gamow’s Theory of Alpha DecayAlpha decay theory using computational tools
REFERENCES
Encyclopædia Britannica. Available in: https://www.britannica.com/science/alpha-decay. Access in: 30/09/2018.
Encyclopædia Britannica. Available in: https://www.britannica.com/biography/George-Gamow. Access in: 30/09/2018.
Wikipedia. Available in: https://en.wikipedia.org/wiki/Alpha_decay. Access in: 30/09/2018.


























0 comments
Sign in or create a free account